 The atomic radius is determined entirely by the electrons: The size of the atomic nucleus is measured in femtometres, 100,000 times smaller than the cloud of electrons. 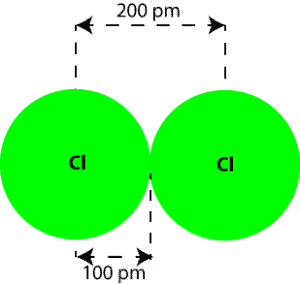
In the latter case, which is the approach adopted here, it should also include ionic radius, as the distinction between covalent and ionic bonding is itself somewhat arbitrary. The term "atomic radius" itself is problematic: it may be restricted to the size of free atoms, or it may be used as a general term for the different measures of the size of atoms, both bound in molecules and free. The value assigned to the radius of a particular atom will always depend on the definition chosen for "atomic radius", and different definitions are more appropriate for different situations. It does not store any personal data.Atomic radius, and more generally the size of an atom, is not a precisely defined physical quantity, nor is it constant in all circumstances. The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. The cookie is used to store the user consent for the cookies in the category "Performance". This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. The cookies is used to store the user consent for the cookies in the category "Necessary". The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". The cookie is used to store the user consent for the cookies in the category "Analytics". These cookies ensure basic functionalities and security features of the website, anonymously. Necessary cookies are absolutely essential for the website to function properly. (A) Be Removal of electron is easier due to increased atomic radius and increase in number of shells down the group. 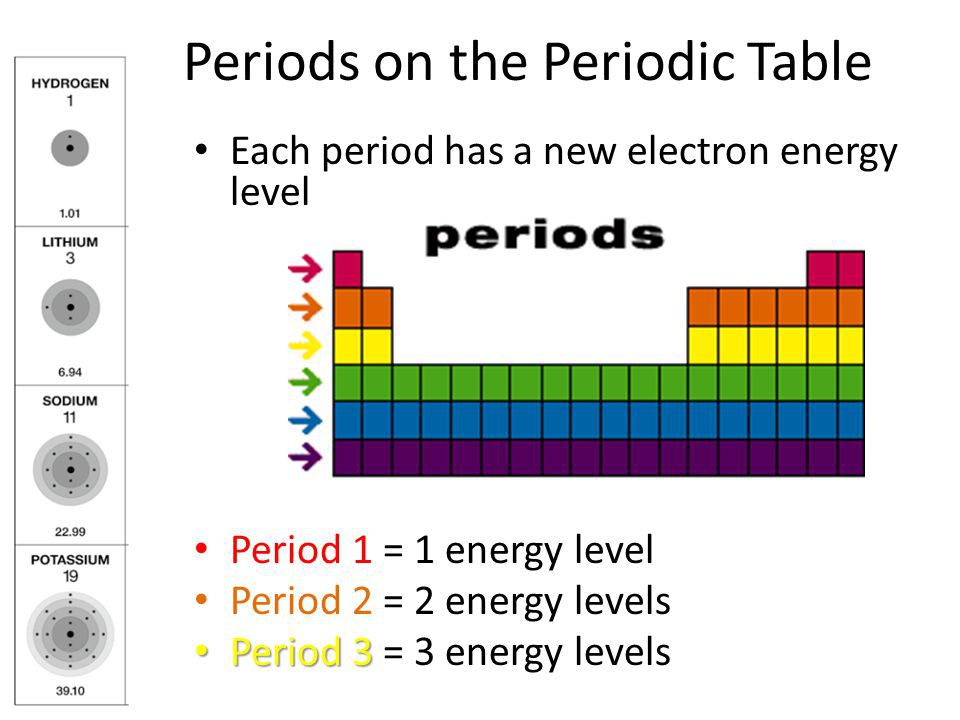
What is the correct decreasing order of atomic radius of the following elements * 1 point? Which of the following correctly represents the decreasing order of atomic radii of the following elements?Įxplanation: (a)order is: Li. Hence, the correct order of increasing ionic radii is Mg2+. The electron cloud expands and the size of negative ion becomes more. When a negative ion is formed, one or more electrons are added to an atom and the effective nuclear charge is reduced. What is the correct decreasing order of ionic radii of following ions n3 O2 F Na+ Mg2+? Because of these two trends, the largest atoms are found in the lower left corner of the periodic table, and the smallest are found in the upper right corner (Figure 3.2. In the periodic table, atomic radii decrease from left to right across a row and increase from top to bottom down a column. How do you order atoms in decreasing size? 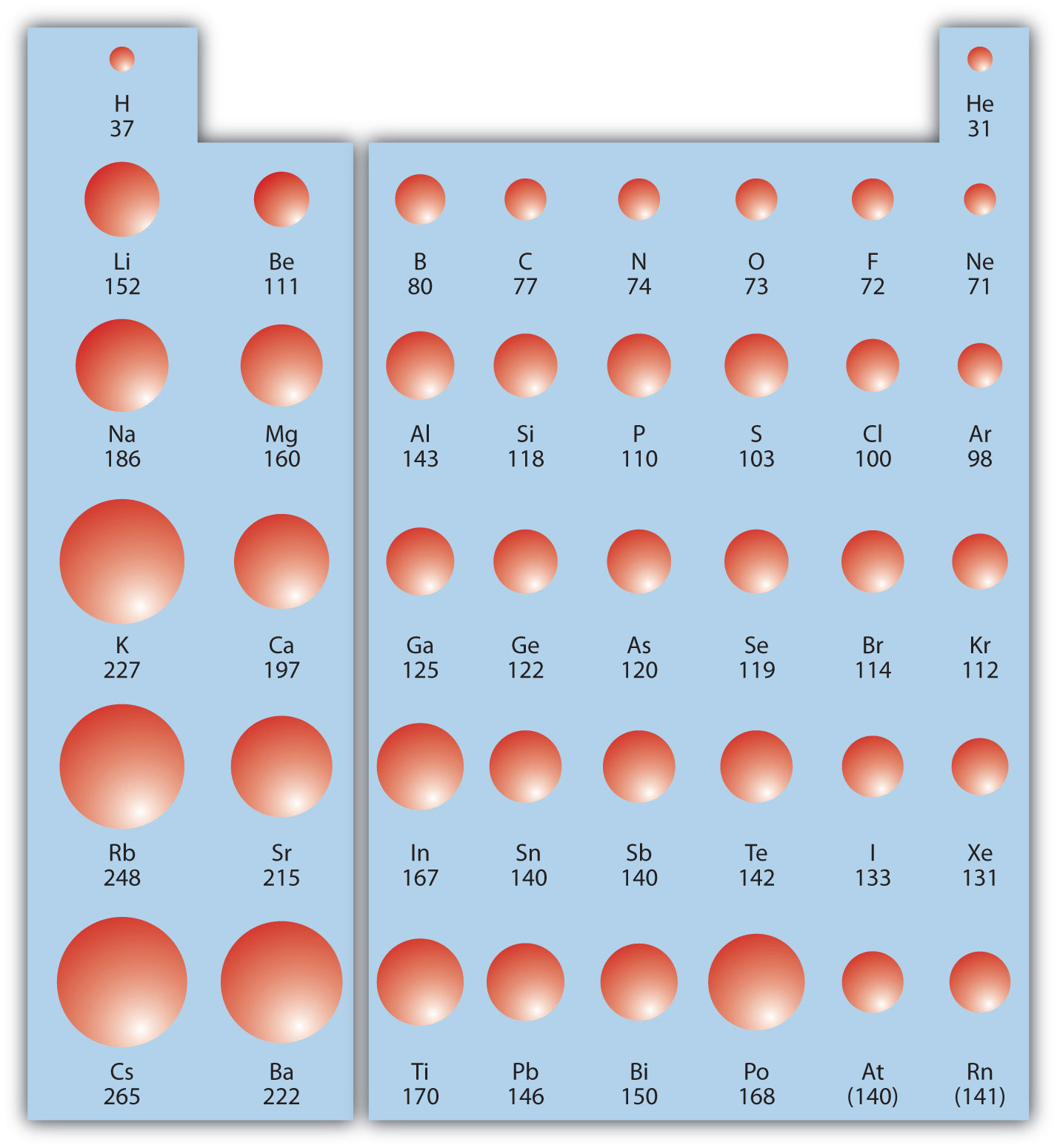
The number of electrons remains the same creating a larger Coulombic attraction and decreasing the radii. 
Which of the following best explains why atomic radius decreases left to right across a period?Īs you move across the period, the number of protons increases, which increases the nuclear charge. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
